Allopurinol and Azathioprine Interaction: Risks and Safety Guidelines

Azathioprine Dose Adjustment Estimator
Estimated Adjusted Dose:
Based on a 75% reduction (25% of original dose).
- Weekly CBC for Month 1
- Bi-weekly CBC for Months 2-3
- Monthly CBC long-term
Enter the current dose to see the calculated adjustment.
Imagine a scenario where a simple prescription for joint pain turns into a life-threatening medical emergency. It sounds like a plot from a medical drama, but for some patients, it is a stark reality. When Allopurinol is a medication used to lower uric acid levels to treat gout is prescribed to someone already taking Azathioprine, an immunosuppressant used for organ transplants and inflammatory bowel disease, the result can be a catastrophic drop in white blood cells. This is not just a mild side effect; it is one of the most dangerous drug-drug interactions in modern medicine.
The core problem is that these two drugs fight over the same metabolic pathway. If you are taking these medications without strict specialist supervision, you are risking severe bone marrow toxicity. This guide explains why this happens, the warning signs you need to watch for, and the very specific protocols doctors use when this combination is absolutely necessary.
Why Allopurinol and Azathioprine Clash
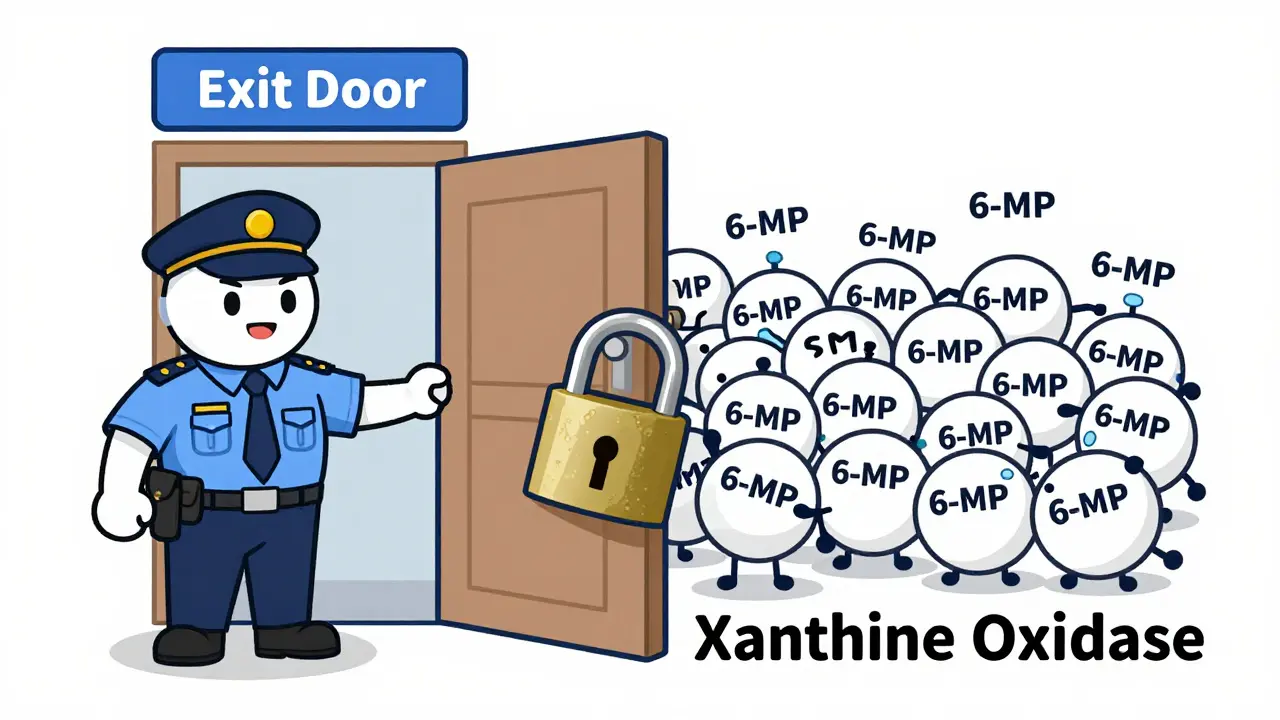
To understand the danger, we have to look at how your body processes medication. Azathioprine is a pro-drug. Once it enters your system, it breaks down into an active metabolite called 6-mercaptopurine (6-MP). Your body then uses an enzyme called Xanthine Oxidase to break down 6-MP into an inactive form, preventing it from building up to toxic levels.
Here is where the trouble starts. Allopurinol is specifically designed to block that exact same enzyme. By inhibiting Xanthine Oxidase, allopurinol essentially closes the exit door for 6-MP. This creates a "shunting" effect, where the drug is forced down a different metabolic path, increasing the levels of active metabolites in your blood by up to four times. When these levels spike, they begin to attack your own bone marrow, stopping the production of the very cells your body needs to survive.
The Danger Zone: Bone Marrow Suppression
When the interaction hits, it leads to Myelosuppression, a condition where bone marrow activity is decreased, resulting in fewer blood cells. This isn't a gradual decline; it can be a rapid crash. In documented clinical cases, patients have seen their white blood cell counts plummet to dangerous levels (as low as 1.1 × 10³/mm³), leaving them completely defenseless against infection.
You might notice symptoms that seem unrelated at first. Extreme fatigue, frequent bruising, or a sudden fever are all red flags. Because the bone marrow is being suppressed, your body stops producing enough red blood cells (leading to severe anemia) and platelets (leading to uncontrolled bleeding). In one historical case, a heart transplant patient required emergency blood transfusions and specialized growth factor therapy just to survive the interaction after being given allopurinol for suspected gout in his wrist.
| Metabolite/Enzyme | Normal Function/Level | Effect of Allopurinol | Clinical Result |
|---|---|---|---|
| Xanthine Oxidase | Degrades 6-MP | Blocked/Inhibited | 6-MP builds up in blood |
| 6-TGNs | Therapeutic levels | Increased significantly | Bone marrow toxicity |
| 6-MMP | Can be hepatotoxic | Reduced (up to 70%) | Lower liver toxicity |
When is the Combination Actually Used?
You might be wondering: if it is so dangerous, why would any doctor ever do this? The answer lies in a specific group of people called "thiopurine shunters." These are patients, often those with Inflammatory Bowel Disease (IBD), whose bodies naturally produce too much 6-MMP (the liver-toxic version) and not enough 6-TGN (the healing version) from azathioprine.
In these rare cases, a specialist might use a tiny dose of allopurinol to intentionally block the "shunting" to the liver and force the medication toward the therapeutic pathway. This can help patients achieve steroid-free remission and improve mucosal healing in the gut. However, this is a high-wire act. It is only done in academic medical centers or specialized IBD clinics-never in a general primary care setting.

Safe Prescribing and Monitoring Protocols
If a specialist decides that the benefits outweigh the risks, they don't just "add" allopurinol to the mix. They follow a strict safety protocol to prevent lethal toxicity. The first rule is a massive dose reduction. Typically, the dose of Azathioprine must be cut to about 25% of the standard amount (dropping from roughly 2.0 mg/kg/day to about 0.5 mg/kg/day).
Monitoring is the only way to keep the patient safe. The standard protocol includes:
- Baseline Tests: Full blood count and liver function tests before the first dose.
- Weekly Blood Work: Complete blood counts (CBC) every single week for the first month.
- Bi-weekly Checks: Monitoring every two weeks for the following two months.
- Long-term Maintenance: Monthly blood tests indefinitely to ensure the bone marrow remains healthy.
Doctors also track specific metabolite levels. They aim for therapeutic targets (230-450 pmol/8×10⁸ RBCs for 6-TGN) while keeping 6-MMP levels below 5,700 pmol/8×10⁸ RBCs to avoid damaging the liver.
Alternatives for Gout and Immunosuppression
Because the risk of allopurinol and azathioprine interaction is so high, the best course of action is usually to find an alternative. There are other ways to manage gout and autoimmune conditions that don't carry the risk of total bone marrow failure.
For gout, physicians may look toward Febuxostat or Pegloticase. These medications lower uric acid through different mechanisms that don't interfere with the metabolism of thiopurines. On the other hand, if a patient cannot tolerate azathioprine due to shunting or toxicity, they might switch to Methotrexate or modern biologic therapies (like TNF inhibitors), which provide similar immunosuppression without the same complex metabolic dangers.
Can I take allopurinol if I am on azathioprine?
In general, no. This combination should be avoided because allopurinol blocks the enzyme needed to clear azathioprine's active metabolites. This can lead to life-threatening bone marrow suppression. It should only be attempted under the direct supervision of a specialist who can reduce your dose by 75% and monitor your blood weekly.
What are the early warning signs of this interaction?
Watch for signs of low blood counts: unusual bruising or bleeding, extreme fatigue, pale skin, and recurrent infections or fever. If you notice these while taking both medications, seek emergency medical care immediately.
Why does the dose of azathioprine need to be reduced?
Because allopurinol stops the body from breaking down the active metabolite 6-MP, the drug stays in your system much longer and at higher concentrations. A standard dose becomes effectively a "massive overdose," which is why the dose must be lowered to 25-33% of the normal amount to prevent toxicity.
Is this interaction the same for 6-mercaptopurine?
Yes. Azathioprine is converted into 6-mercaptopurine (6-MP) in the body. Therefore, allopurinol interacts with 6-MP in the exact same way, leading to the same risks of myelosuppression.
What is a "thiopurine shunter"?
A shunter is a person whose body processes azathioprine incorrectly, producing too many toxic metabolites that damage the liver (6-MMP) and too few therapeutic metabolites (6-TGN). In these specific cases, allopurinol is sometimes used as a tool to "re-route" the metabolism to make the drug work better.

Clint Humphreys
April 14, 2026 AT 13:59It is honestly just so funny how they present this as a "medical emergency" when you realize the pharmaceutical giants probably knew about this interaction decades ago but just didn't want to put it in the fine print because that would mess with the profit margins of both drugs. I mean just think about it for a second because if the system is designed to help us why is it that the most dangerous combinations are the ones that are just a couple of prescriptions away from a primary care doctor who probably hasn't read a textbook since 1998 and is just clicking buttons on a screen while the actual chemistry of our bone marrow is being dismantled by a "shunting effect" that sounds more like a subway accident than a biological process. It is all just one big game of trial and error with our lives as the pawns but hey at least the doctors get a nice bonus for the "specialist supervision" they provide in those fancy academic centers where they can play god with 25% dose reductions and pretend they are saving us from the very disaster their own industry created in the first place.
Shaylia Helland
April 15, 2026 AT 09:18this is honestly such a scary thought just imagining your white blood cells just disappearing because of a mix up but i guess it makes sense why they have to be so careful about it especially with the whole enzyme thing and the way the body just tries to find another way to get rid of the stuff which ends up being way worse in the long run and i just feel for the people who have to deal with the anxiety of weekly blood tests just to make sure they are still okay while fighting something like ibd which is already a total nightmare to live with day to day
Haley Moore
April 15, 2026 AT 17:39Ugh please tell me someone else finds it absolutely exhausting that we even have to read these guides because the medical system is basically a joke at this point 🙄 like imagine trusting a GP with this? Absolute chaos. I could never 💅
Tabatha Pugh
April 16, 2026 AT 16:05Actually, most people don't even realize that Xanthine Oxidase isn't just for these drugs but plays a huge role in purine metabolism generally. You're barely scratching the surface here. I've seen cases where patients didn't even have gout but were given allopurinol for other reasons and the result was exactly the same systemic collapse. It's practically basic pharmacology if you've actually studied it.
john chiong
April 16, 2026 AT 22:40absolute carnage in the marrow if you screw this up. just an utter disaster waiting to happen when doctors play fast and loose with the chemistry of the human body. pure madness
David Snyder
April 18, 2026 AT 20:34It is great that there are alternatives like Febuxostat for people who need to manage both conditions. It gives a lot of hope to know that science has found a way around these dangerous interactions so patients can stay healthy and safe.
melissa mac
April 20, 2026 AT 06:19I appreciate the reminder to keep a close eye on symptoms. It's so important for us to support one another in navigating these complex treatments and making sure everyone feels empowered to speak up if something feels off with their health.
Ikram Khan
April 20, 2026 AT 17:46Wow this is absolutely wild!! 😱 I had no idea a dose reduction could be that extreme just to keep someone safe! It really shows how powerful these medications are when they clash! 💊💥